Knowledge Base
Enterprise Resource Planning (ERP) system overview
Enterprise Resource Planning (ERP) in the Biotech Industry: Structure, History, and Integration with LIMS and ELN
The biotechnology and biopharms industries operate at the intersection of science, manufacturing, quality, and regulation. Managing this complexity requires robust digital systems that ensure compliance, traceability, efficiency, and scalability. Enterprise Resource Planning (ERP) systems serve as the operational backbone of biotech organizations, integrating business processes across departments while interfacing with scientific systems such as Laboratory Information Management Systems (LIMS) and Electronic Laboratory Notebooks (ELN).
What Is ERP?
Enterprise Resource Planning (ERP) is an integrated software platform that manages and automates an organization’s core business processes using a centralised database.
In biotech, ERP systems support:
-
Manufacturing and batch control
-
Supply chain and inventory management
-
Quality management and compliance
-
Financials and cost tracking
-
Human resources and training records
ERP ensures that data flows seamlessly between departments, reducing silos and enabling real-time decision-makin
Historical Evolution of ERP
1. MRP (1960s–1970s)
-
Material Requirements Planning (MRP) focused on inventory control and production planning.
-
Primarily used in manufacturing industries.
-
Limited to materials and scheduling.
2. MRP II (1980s)
-
Expanded to include capacity planning, finance, and workforce considerations.
-
Introduced feedback loops between production and planning.
3. ERP Emergence (1990s)
-
Integrated finance, HR, procurement, manufacturing, and logistics.
-
Introduced centralised databases and modular architectures.
-
Became enterprise-wide systems rather than manufacturing-only tools.
4. Modern ERP (2000s–Present)
-
Cloud-based, API-driven, and scalable.
-
Supports regulatory frameworks such as GMP, GxP, FDA 21 CFR Part 11.
-
Designed to integrate with specialised systems like LIMS and ELN.
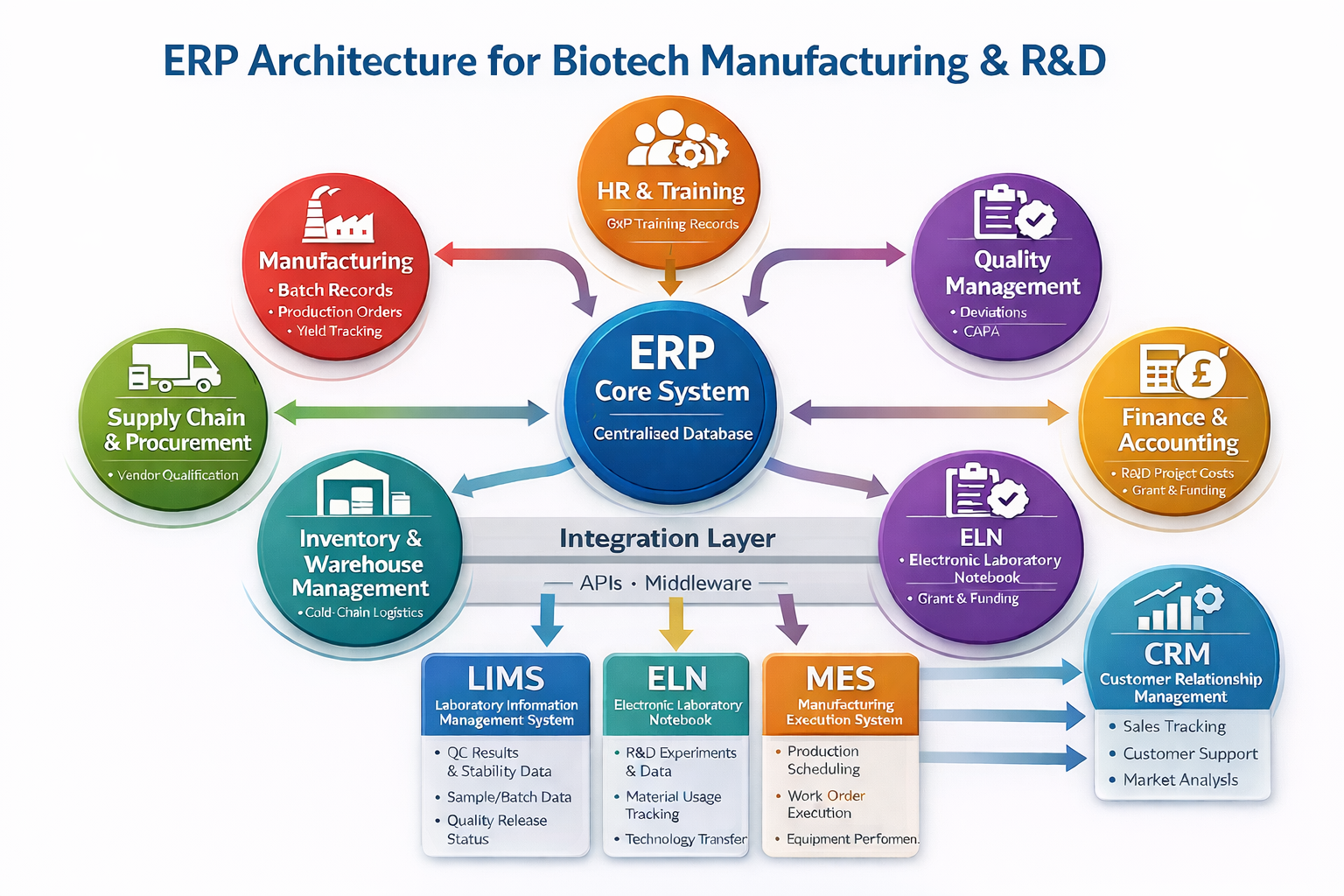
Core Structure of ERP Systems
In biotech organisations, ERP systems function as the operational and compliance backbone that connects research activities with regulated manufacturing and commercial operations. Unlike traditional ERP implementations, biotech ERP must support batch-based manufacturing, strict quality controls, traceability, and regulatory compliance, while also aligning closely with research and development workflows. This dual role makes ERP a critical bridge between scientific innovation and scalable, compliant production.
1. Central Database
At the heart of any Enterprise Resource Planning (ERP) system lies the central database, which serves as the single source of truth for the entire organisation. All business units; finance, manufacturing, quality, supply chain, and human resources access and update data from this unified repository. In the biotech industry, this centralised data model is especially critical because it ensures consistency across regulated processes, supports complete audit trails, and enables compliance with stringent regulatory standards such as GMP and GxP. By eliminating data silos, the central database allows real-time visibility into operations and supports accurate reporting and inspections.
2. Core ERP Modules
Built around this central database are the core ERP modules, each designed to manage a specific functional area while remaining tightly interconnected. The Finance and Accounting module tracks costs, budgets, grant funding, and capital expenditures, which is essential in research-driven biotech organisations. The Supply Chain module manages raw material sourcing, vendor qualification, and cold-chain logistics to ensure material availability and integrity. The Manufacturing module supports batch records, production orders, and scheduling, enabling controlled and traceable production processes. The Quality Management System (QMS) module handles deviations, CAPA, audits, and change control, forming the backbone of regulatory compliance. The Inventory Management module ensures accurate lot tracking, stock visibility, and expiry management, while the HR and Training module maintains employee records, role-based training, and compliance qualifications.
3. Integration Layer
A defining feature of biotech ERP architecture is its integration layer, which connects ERP with specialised scientific and execution systems. ERP integrates with LIMS to receive quality control test results, stability data, and batch release status, enabling informed disposition decisions. Integration with ELN allows ERP to link R&D experiments with project budgets, material consumption, and technology transfer activities. In manufacturing environments, ERP also interfaces with MES to synchronize production execution data, ensuring consistency between planned and executed processes. These integrations create a digital thread that spans discovery, development, scale-up, and commercial manufacturing.
Overall, ERP architecture in biotech manufacturing and R&D supports regulatory compliance, operational efficiency, and scientific continuity. By tightly integrating ERP with laboratory and execution systems, biotech/biopharma organisations can reduce manual data handling, accelerate technology transfer, improve audit readiness, and ensure that innovative research can be reliably transformed into safe, high-quality products.
Integration of ERP with LIMS
The integration of an ERP system with a LIMS is a critical component of the digital architecture in regulated biotech environments, particularly for quality control and manufacturing operations. LIMS is responsible for managing laboratory-centric activities such as sample lifecycle tracking, analytical test execution, stability studies, and QC/QA workflows, while ERP governs enterprise-level processes including batch management, inventory, quality disposition, and financial control. Through system integration, ERP initiates the creation of samples and batches in LIMS based on production or procurement events, ensuring alignment between manufacturing execution and laboratory testing. LIMS, in turn, transmits analytical results, stability outcomes, and quality status back to ERP, enabling automated updates to batch release, inventory availability, and disposition decisions. This bidirectional data exchange eliminates manual transcription, reduces data integrity risks, and ensures end-to-end traceability from raw materials through finished goods. By synchronising laboratory results with enterprise quality and supply chain processes in near real time, ERP–LIMS integration supports compliant, timely quality release decisions and strengthens audit readiness in GxP-regulated biotech operations.
Integration of ERP with ELN
The integration of an ERP system with an ELN is a foundational element of a digitally connected biotech R&D and manufacturing environment. ELN systems are designed to capture, structure, and retain scientific knowledge generated during research activities, including experimental protocols, raw and processed data, calculations, observations, and intellectual property records. These systems operate within a controlled, time-stamped, and versioned framework to ensure data integrity and reproducibility, which are essential for regulatory submissions and future technology transfer. While ELN focuses on the scientific and experimental context, it operates largely independently of enterprise-level controls such as financial governance, material planning, and resource allocation, which are managed by ERP.
ERP–ELN integration bridges this gap by enabling structured data exchange between research execution and enterprise operations. R&D projects defined within ERP are linked to corresponding experiments and studies in ELN, allowing experimental activities to be associated with approved cost centres, budgets, and funding sources. Material master data maintained in ERP such as raw materials, reagents, and consumables is synchronised with ELN to enable accurate tracking of material usage during experiments, ensuring consistency between laboratory consumption and enterprise inventory records. This integration is particularly critical during technology transfer, where process knowledge, formulations, and experimental outcomes captured in ELN are referenced by ERP-driven manufacturing planning and costing activities. By aligning scientific data with enterprise controls, ERP–ELN integration provides full visibility into R&D expenditure, supports controlled scale-up from laboratory to pilot and commercial manufacturing, and enables seamless collaboration between research scientists, manufacturing, quality, and finance teams within a compliant, traceable digital framework.