Knowledge Base
Laboratory Information Management System (LIMS) overview
Laboratory Information Management Systems (LIMS): Overview and Architecture
Laboratory Information Management Systems (LIMS) have become a critical component of modern laboratory operations across research, analytical, clinical, and industrial environments. As laboratories generate increasing volumes of complex data, LIMS provides the foundation for managing samples, workflows, results, compliance, and collaboration efficiently and securely. Beyond simple data storage, modern LIMS platforms act as central hubs that connect laboratory processes, instruments, and business systems into a unified digital ecosystem.
What Is a LIMS?
A LIMS is a software platform designed to manage laboratory information throughout its lifecycle. It supports the tracking of samples from receipt through analysis and reporting, ensures data itegrity, enforces standard operating procedures (SOPs), and enables regulatory compliance. By automating routine tasks and centralising laboratory data, LIMS reduces manual errors, improves traceability, and enhances overall laboratory productivity.
Core functions of a LIMS typically include sample management, test and method execution, results capture, quality control, audit trails, reporting, and user access management. Modern LIMS solutions are highly configurable, allowing laboratories to adapt the system to their specific scientific, operational, and regulatory requirements.
The Evolution of LIMS
Early LIMS implementations were often rigid, on-premise systems focused primarily on sample tracking. Today’s LIMS platforms have evolved into flexible, cloud-enabled solutions that support complex workflows, real-time data exchange, and integration with enterprise systems such as ERP, ELN, and CRM. This evolution reflects the growing need for scalability, interoperability, and data-driven decision-making within scientific organisations.
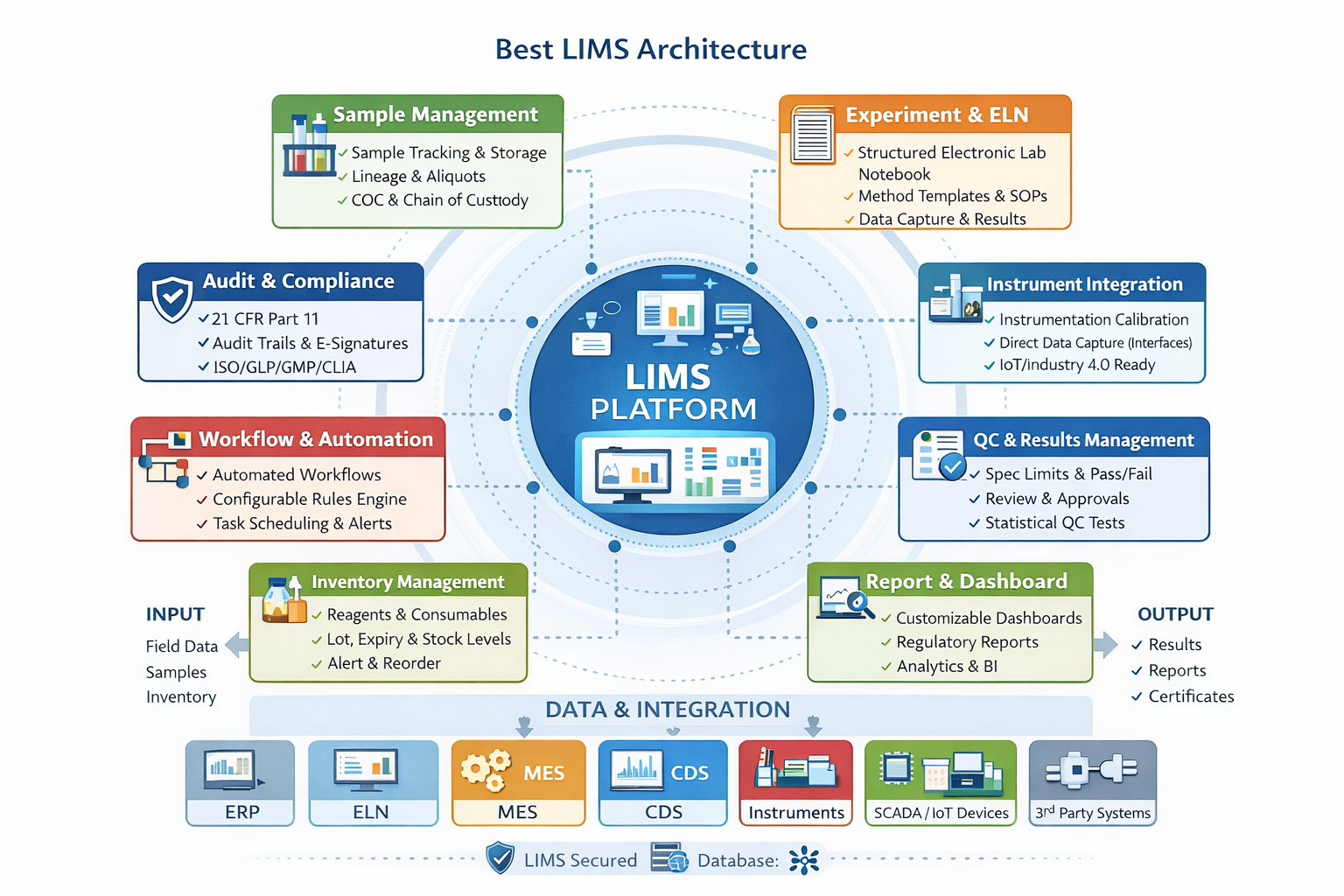
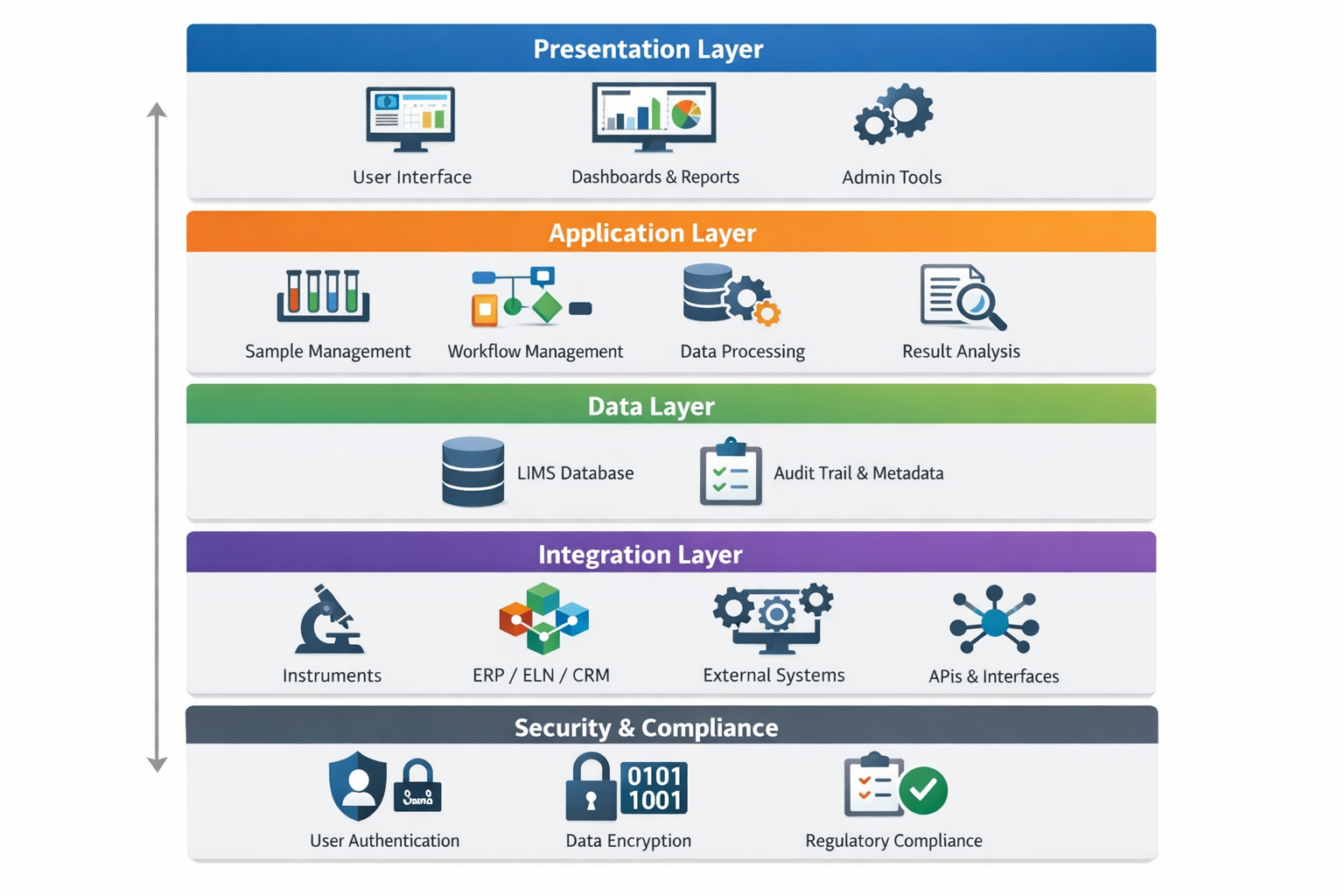
LIMS Architecture Overview
LIMS architecture defines how system components are structured, how data flows between them, and how the platform integrates with external systems. A well-designed architecture ensures performance, scalability, security, and long-term maintainability.
1. Presentation Layer (User Interface)
The presentation layer provides the user-facing interface through which laboratory personnel interact with the system. Modern LIMS platforms typically use web-based interfaces that are intuitive, role-based, and accessible across devices. This layer supports dashboards, workflows, data entry forms, reporting views, and administrative configuration tools.
2. Application Layer (Business Logic)
The application layer contains the core business logic that governs laboratory workflows and rules. This includes sample lifecycle management, method execution, validation rules, approvals, calculations, and compliance enforcement. By separating business logic from the user interface, the system remains flexible and easier to configure or extend as laboratory needs evolve.
3. Data Layer (Database and Storage)
The data layer is responsible for storing all laboratory information, including samples, results, metadata, audit trails, and configuration data. Robust database design is critical to ensure data integrity, traceability, and performance. Modern LIMS solutions often support structured and semi-structured data, enabling efficient handling of both numerical results and unstructured scientific content.
4. Integration Layer
The integration layer enables communication between LIMS and external systems such as laboratory instruments, ELNs, ERP platforms, quality management systems (QMS), and reporting tools. This layer typically uses APIs, web services, or message queues to ensure secure, reliable data exchange and to support automation across the laboratory and enterprise environment.
5. Security and Compliance Layer
Security is embedded throughout the LIMS architecture. This includes authentication, role-based access control, data encryption, audit logging, and compliance with regulatory standards such as GLP, GMP, ISO, and FDA 21 CFR Part 11. A strong security framework ensures data confidentiality, integrity, and accountability.
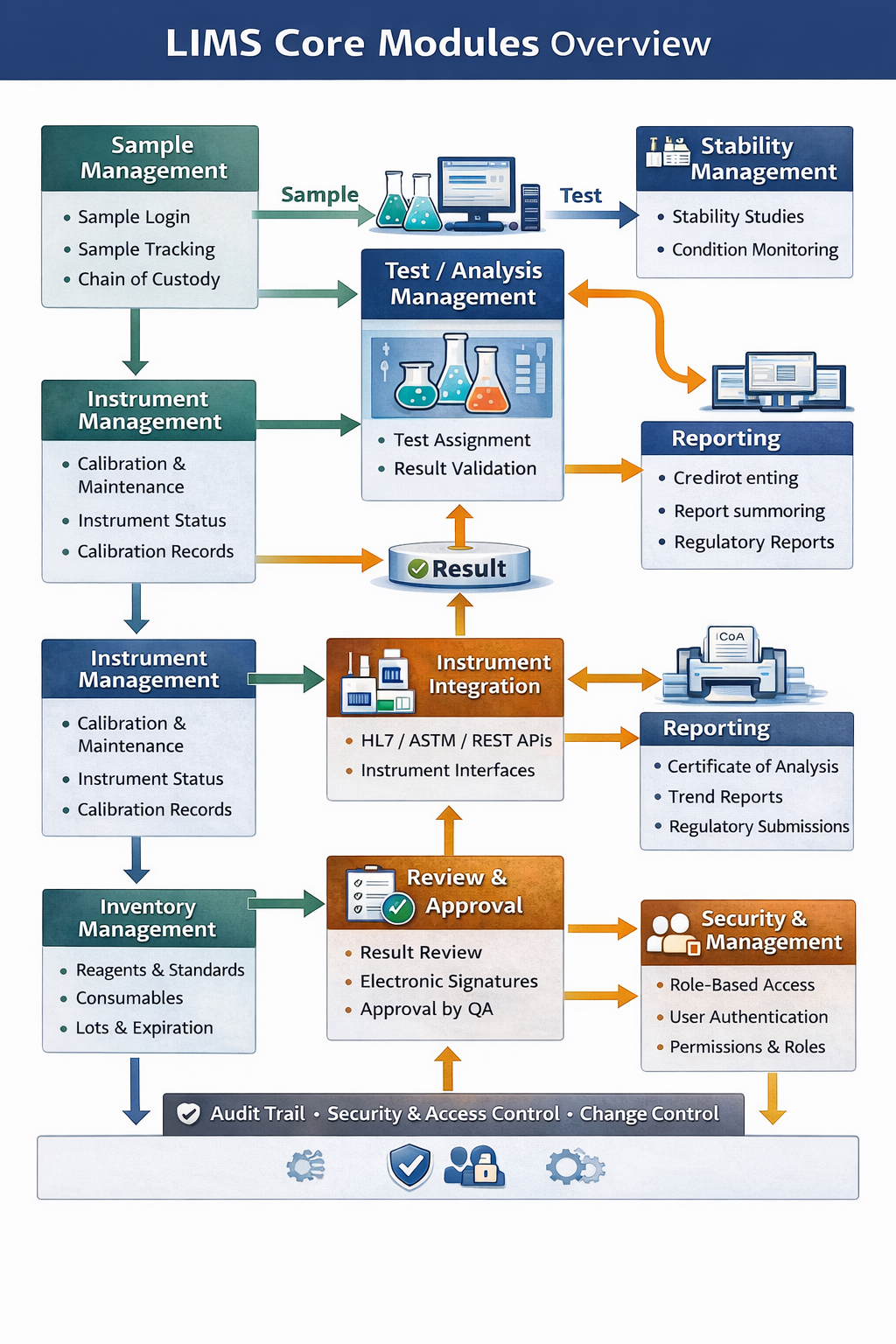
Core Components of an Advanced LIMS and Their Structure
An advanced LIMS is built as a modular, layered platform designed to support complex laboratory workflows, high data volumes, regulatory compliance, and enterprise integration. Its structure ensures scalability, flexibility, and long-term sustainability.
1. Sample & Asset Management Core
Purpose:
Manages the complete lifecycle of samples, materials, and laboratory assets.
Key Functions:
- Sample registration, barcoding, and tracking
- Chain of custody and status management
- Sample storage and location tracking
- Reagents, standards, and consumables management
Structure:
- Centralised sample master data model
- State-based lifecycle engine
- Relationship mapping between samples, tests, and batches
2. Instrument Management Module
Purpose:
Ensures only qualified, calibrated, and maintained instruments are used to generate laboratory data.
Key Functions:
- Hold instrument metadata (Manufacturer, model, serial number, location, ownership and status)
- Calibration schedules and due dates managment
- Maintenance management including corrective and preventative schedules
- Service vendor documentation
- Instrument lifecycle tracking (IO, OQ,PQ)
- Integration with testing (ELN)
Structure:
- Centralised instrument master data model
- Event-driven process execution
- Orchestration and monitoring tools
- Role-based access and segregation of duties
2. Inventory Management Module
Purpose:
Controls and tracks reagents, standards, consumables, and materials used in testing to ensure accuracy, traceability, and compliance.
Key Functions:
- Reagent and standard master data (CoA, supplier, manufacturer and storage conditions)
- Lot/batch number control and tracking
- Inventory Quantity Tracking (stock levels)
- Usage Traceability
- Integration with Testing
Structure:
- Rule-based workflow engine
- Visual workflow configuration layer
- Event-driven process execution
4. Workflow & Process Orchestration Engine
Purpose:
Defines, executes, and enforces laboratory workflows.
Key Functions:
- Configurable workflows and SOP enforcement
- Task assignment and approvals
- Conditional logic and exception handling
- Parallel and sequential process execution
Structure:
- Rule-based workflow engine
- Centralised inventory master data model
- Event-driven process execution
5. Test, Method & Result Management
Purpose:
Controls analytical methods and manages test execution and results.
Key Functions:
- Method definitions and version control
- Parameter specifications and calculations
- Results capture and validation
- Out-of-specification (OOS) handling
Structure:
- Method repository with versioning
- Calculation and validation engine
- Results data model linked to samples and workflows
6. Data Management & Persistence Layer
Purpose:
Ensures secure, structured, and traceable data storage.
Key Functions:
- Storage of structured and semi-structured data
- Metadata and audit trail capture
- Data integrity and traceability
- High-performance querying and reporting
Structure:
- Relational and/or hybrid databases
- Immutable audit logs
- Configurable data schemas
7. Instrument & System Integration Layer
Purpose:
Enables automated data exchange with laboratory instruments and enterprise systems.
Key Functions:
- Instrument data capture (file-based or real-time)
- Integration with ELN, ERP, QMS, CRM
- API and message-based communication
- Data transformation and validation
Structure:
- REST and event-based APIs
- Integration adapters and connectors
- Message queues and middleware services
8. Reporting, Analytics & Intelligence Layer
Purpose:
Transforms laboratory data into actionable insights.
Key Functions:
- Standard and custom reporting
- Real-time dashboards
- Trend analysis and KPIs
- Regulatory and management reports
Structure:
- Reporting engine
- Analytics and aggregation services
- Visualisation and dashboard layer
9. Configuration & Customisation Framework
Purpose:
Allows system adaptation without custom code.
Key Functions:
- Configurable data models and workflows
- Role-based UI configuration
- Business rule management
- Multi-tenant configuration support (SaaS)
Structure:
- Metadata-driven configuration engine
- UI schema definitions
- Versioned configuration repository
10. Security, Compliance & Governance Layer
Purpose:
Ensures data protection and regulatory compliance.
Key Functions:
- User authentication and authorisation
- Electronic signatures
- Audit trails and change control
- Compliance with GLP, GMP, ISO, 21 CFR Part 11
Structure:
- Role-based access control (RBAC)
- Policy enforcement services
- Compliance audit framework
11. Deployment & Infrastructure Layer
Purpose:
Supports scalability, availability, and performance.
Key Functions:
- Cloud-native or hybrid deployment
- High availability and disaster recovery
- Performance monitoring and logging
- Automated updates and scaling
Structure:
- Containerised services (microservices)
- Orchestration and monitoring tools
- Secure cloud infrastructure
Differences Between LIS and LIMS
Laboratory information systems play a critical role in modern laboratories by ensuring data accuracy, traceability, and efficiency. Two commonly used systems are the Laboratory Information System (LIS) and the Laboratory Information Management System (LIMS). Although these systems are sometimes confused or used interchangeably, they are fundamentally different in purpose, design, and regulatory focus. Understanding the differences between LIS and LIMS is essential for selecting the right system for a laboratory’s operational and compliance needs.
At a high level, the primary distinction between LIS and LIMS lies in what they manage. An LIS is designed to support patient-centric clinical testing, while a LIMS is designed to manage sample-centric laboratory operations, particularly in regulated and quality-driven environments.
An LIS is most commonly used in hospitals, clinical diagnostic laboratories, and pathology labs. Its main function is to manage patient test orders, track samples associated with patients, and report diagnostic results to clinicians and electronic medical record (EMR) systems. The core data model of an LIS revolves around the patient. A typical workflow begins with a physician ordering a test for a patient, followed by sample collection, test execution, result verification, and result reporting. Speed, accuracy, and timely communication of results are the primary performance drivers of an LIS. As a result, LIS systems are optimised for high throughput, rapid turnaround times, and seamless integration with hospital information systems using standards such as HL7.
In contrast, a LIMS is used primarily in pharmaceutical, biotechnology, food and beverage, environmental, manufacturing, and research laboratories. These laboratories are generally not patient-focused but are instead concerned with samples, methods, instruments, and regulatory compliance. The LIMS data model centers on the sample rather than the patient. Samples may originate from production batches, stability studies, environmental monitoring, or research projects. Each sample may undergo multiple tests, methods, and reviews before results are approved and released.
One of the most significant differences between LIS and LIMS is how they handle compliance and data integrity. Clinical laboratories using LIS are typically regulated under standards such as HIPAA and CLIA, which emphasise patient privacy and diagnostic accuracy. While audit trails and access controls may exist, they are often limited to result verification and user authentication. In contrast, LIMS systems are built to meet stringent regulatory requirements such as FDA 21 CFR Part 11, GLP, GMP, GCP, ISO 17025, and ALCOA+ data integrity principles. LIMS architectures enforce full audit trails, electronic signatures, segregation of duties, and system-enforced workflows that prevent unauthorised or undocumented changes to data.
Deployment Models
Modern LIMS platforms can be deployed using different models depending on organisational needs:
- On-premise LIMS offer full infrastructure control but require significant IT resources.
- Cloud-based (SaaS) LIMS provide scalability, lower maintenance overhead, and faster deployment.
- Hybrid architectures combine on-premise and cloud components to meet specific regulatory or operational requirements.
Benefits of a Well-Designed LIMS Architecture
A robust LIMS architecture delivers numerous advantages, including improved laboratory efficiency, enhanced data quality, simplified compliance, and seamless system integration. It enables laboratories to scale operations, adopt new technologies, and respond quickly to changing scientific and regulatory demands.
Why SynthVaults?
SynthVaults LIMS is designed to support modern laboratories by providing a centralised system for managing samples, experiments, data, and workflows throughout the entire laboratory lifecycle. It enables laboratories to efficiently track samples, methods, results, and compliance requirements while ensuring data integrity, traceability, and regulatory readiness.
Built for flexibility and scalability, SynthVaults LIMS caters to research, analytical, and production-focused laboratories by adapting to diverse scientific workflows and operational needs. The system streamlines laboratory operations, reduces manual processes, and improves collaboration by integrating experimental data, instrumentation outputs, and documentation into a single, intuitive platform.
Seamlessly integrated within the broader SynthVaults ecosystem including ERP, ELN, BSS, and CRM SynthVaults LIMS acts as the operational backbone of the laboratory, enabling end-to-end visibility, faster decision-making, and consistent, high-quality scientific outcomes.